NAD+-Utilizing Enzymes
NAD+-Utilizing Enzymes are Critical to Stress Support Pathways in Cancer and Inflammation
NAD+ is a critical stress response molecule. Many cellular stress pathways rely on enzymes that use NAD+ as a co-substrate. There are several categories of NAD+-utilizing enzymes, including:
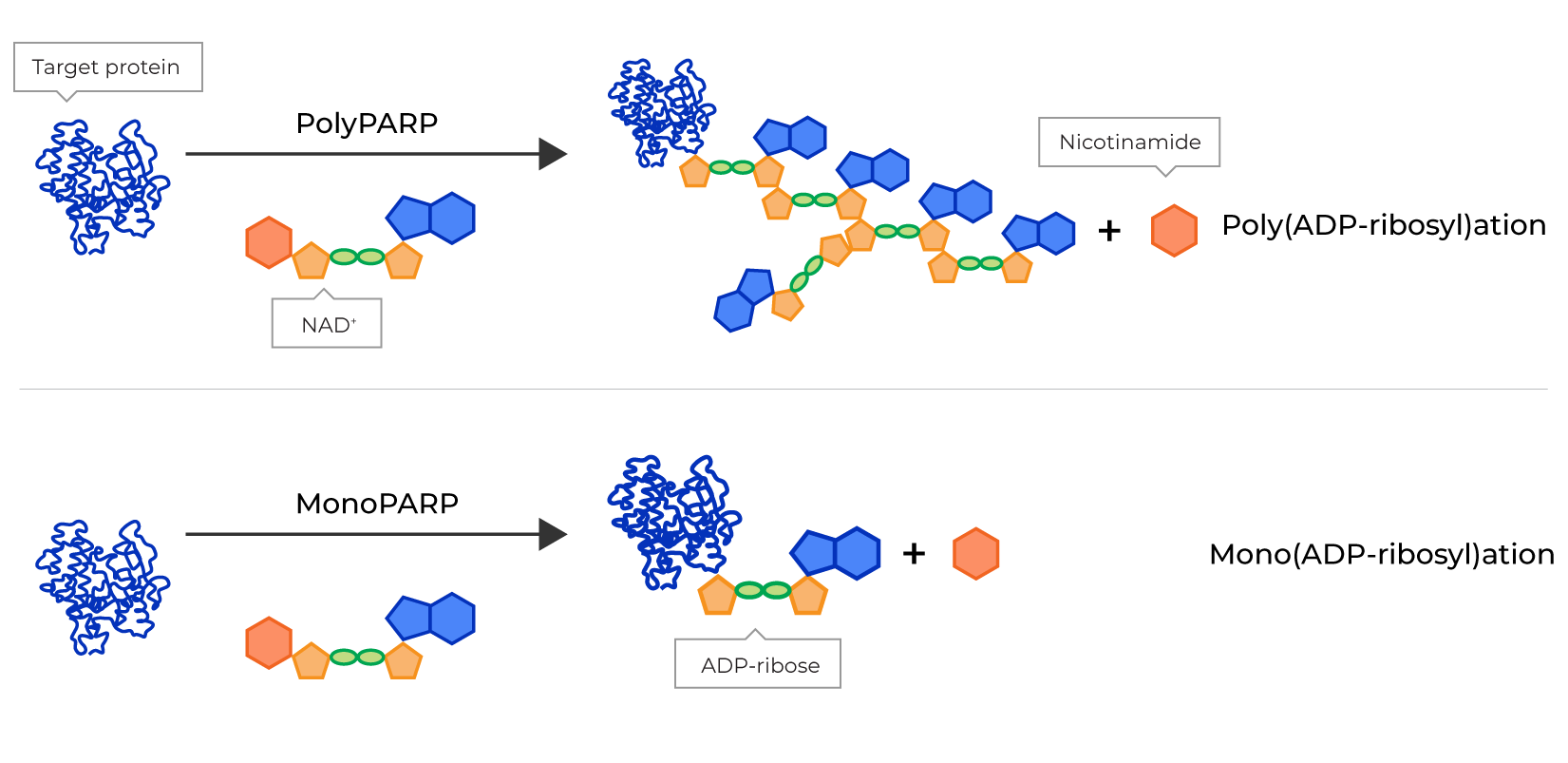
- PARPs, which are a 17-member family of enzymes that vary in structure, cellular location and function but all of which break down NAD+, a co-substrate central to cellular metabolism, and attach ADPR, a part of the NAD+ molecule, to proteins, a process referred to as ADP-ribosylation.
- NAD+ hydrolases, which break down NAD+ into ADPR and nicotinamide. Certain of these enzymes are upregulated in some cancers or in response to treatment with immune checkpoint inhibitors, leading to an immunosuppressive environment for the tumors to grow.
- ARTs, which perform a similar reaction to monoPARPs, by transferring a single molecule of ADPR to proteins in response to different stress. ART1, in particular, has shown higher than normal expression in some cancers.
The PARP Family’s Potential as Novel Therapeutic Targets
The PARP family is divided into two sub-classes: polyPARPs and monoPARPs. PolyPARPs sequentially add many ADPR molecules to proteins, resulting in chains of ADPR that can be hundreds of units long. In contrast, monoPARPs modify proteins by attaching only a single ADPR molecule. Consequently, polyPARPs and monoPARPs represent two separate classes of therapeutic targets as they have diverse protein substrates subject to ADP-ribosylation that perform distinct roles in cell signaling and regulation of protein function. As shown below, Poly(ADP-ribosyl)ation, or PARylation, is the addition of two or more ADPR units that often functions as a scaffold for protein recruitment. Mono(ADP-ribosyl)ation, or MARylation, is the addition of one ADPR unit that acts as a switch to regulate protein function.
NAD+ is a critical molecule in the activation of stress support pathways in cancer cells. There are several categories of enzymes that utilize NAD+ as a co-substrate.